By Obinwanne Chukwuemeka Junior (OD, FAAO, FLVPEI, FSLS) / obinwannejr@gmail.com;
Head Of Cornea, Specialty Contact Lenses and Myopia Control Services, De Lens Ophthalmics Family Eye And Vision Care Centre, Febson Mall, Abuja, Nigeria.
Keratoconus is a bilateral, sometimes asymmetric, non-inflammatory condition of the cornea which results in cornea thinning, ectasia, irregular astigmatism and decreased vision. The prevalence of keratoconus varies depending on the population studied, climate, exposure to risk factors and genetics. It is estimated that in the United States, the prevalence of keratoconus is 0.15%, and is estimated to be 1.5 per 100,000 persons in Finland, 2300 per 100,000 persons in Central India, 0.3 per 100,000 persons in Russia.1 In Africa however, there is a paucity of studies regarding keratoconus.
Considering the fact that keratoconus tends to be associated with allergic eye disease such as vernal keratoconjunctivitis and warm climate, conditions common to Africa, one would expect a plethora of studies regarding keratoconus in Africa, however, a recent meta-analysis of regarding the prevalence of keratoconus in Africa, reviewed only 12 studies which met the criteria for review and estimated the prevalence to be 7.9% in Africa, more in males than females.2
While the number of studies reviewed is minimal, such a prevalence in comparison to other populations begs the question where are the keratoconus patients in Africa? And why are they not presenting to our practices?
As a clinician, managing patients with keratoconus in Africa, these patients are often misdiagnosed as having amblyopia, astigmatism, or corneal scars with little to no proper management expected for a patient with keratoconus before either being referred at late stage of the disease. In most cases, there is limited access to eye care services and the patients resign to the fate of avoidable visual impairment or blindness from the disease. In some cases, however, the patients personally seek better options of care personally and finally end up on the chair of a practitioner who offers management for keratoconus after many unsatisfied visits to numerous eye care providers.
While the current standard diagnosis and classification of keratoconus requires sophisticated equipment like a corneal tomographer in order to determine anterior and posterior corneal curvature and elevation, corneal thickness as well as thinnest pachymetry of the cornea; this device may be out of reach for the average clinician in Africa, leading to either misdiagnosis, or late diagnosis.
There are however other tools which are inexpensive and have been proven effective in diagnosing keratoconus. While these in no way take away the need for standard tools such as the corneal tomographer and slit lamp biomicrosopic signs in diagnosing keratoconus, they may aid the clinician to diagnose the condition, and either manage or refer to the appropriate colleague who offers specialty services for keratoconus.
The tried and trusted streak retinoscope has been shown time and time again to be an effective tool in diagnosing keratoconus. The presence of scissors reflex has been shown to have a sensitivity and specificity of 97.7% and 79.9% in the diagnosis of keratoconus with a positive and negative predictive value of 70.8% and 98.4% respectively.3 With such an impressive statistic, the retinoscope can aid clinicians in identifying this disease and the presence of scissors reflex has even been used in conjunction with keratometry to determine the prevalence of keratoconus. 4

Another inexpensive tool which may serve in identifying keratoconus is the placido disc, while this may be highly qualitative and unreliable in detecting early stages of keratoconus, it is definitely a tool which could be used in large scale screenings in low-income countries to identify keratoconus suspects and refer for further investigation. The change in the shape of the reflected mires from the placido disc, with the rings being closely packed together often indicates corneal steepening and may be indicative of keratoconus.
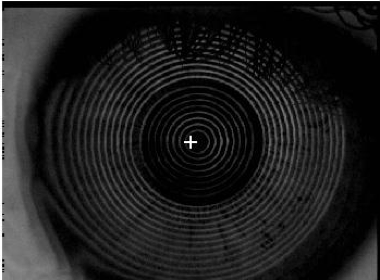
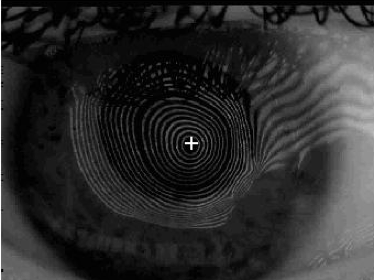
Figure 2: Image of a placido disc reflection on a normal cornea versus a cornea with keratoconus, note the oval shape of the mires and closely packed mires in the second image. Photo courtesy: Dr Obinwanne C. Jr.
The manual keratometer is also an effective tool in diagnosing keratoconus, despite its limitation in being able to provide only anterior corneal curvature data limited to the central 3mm of the cornea with a limited range, it can provide qualitative information on the irregular shape of the cornea. Based on the Collaborative longitudinal Evaluation of keratoconus study (CLEK study) staging of keratoconus based on keratometry, practitioners’ suspicion should be on the alert for keratoconus with keratometry readings above 45D, with >52D being regarded as advanced keratoconus. 5,6
The slit lamp biomicroscope is definitely a versatile tool used in ophthalmic practice, for patients with keratoconus, not only does it aid in the diagnosis, management and follow up, it is also useful in contact lens fitting for these patients. The slit lamp biomicroscopic signs of keratoconus includes prominent corneal nerves, Fleisher’s ring, Vogt’s straie, corneal ectasia and thinning, apical scarring, Rizzuti’s sign and Munson’s sign. 7
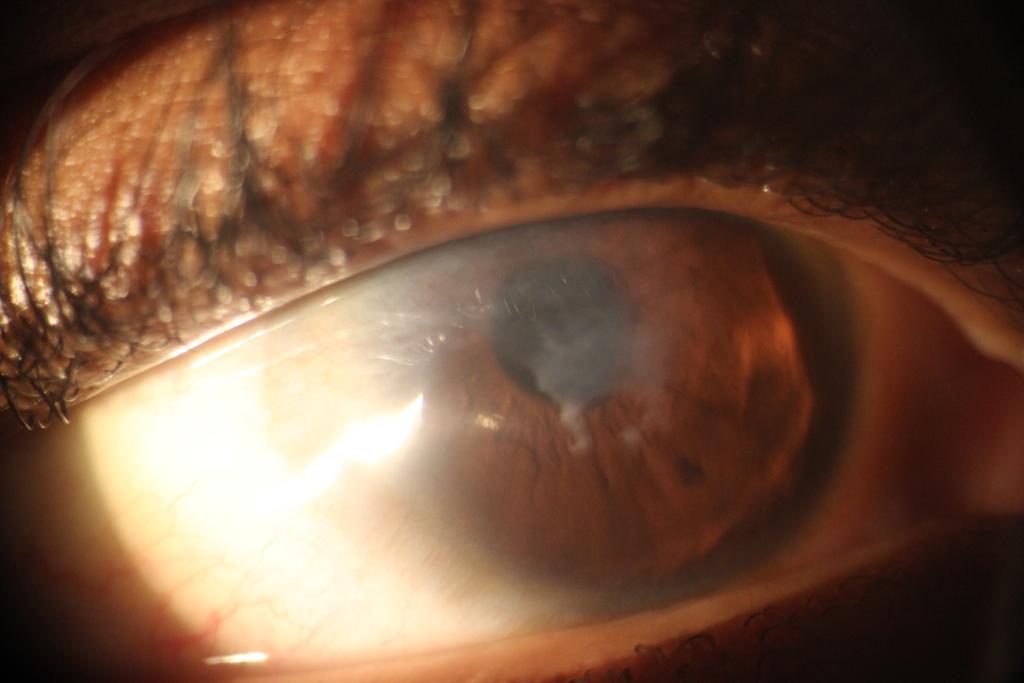
Fleischer’s ring is a faint brownish ring observed in the mid periphery of the cornea, often more visible as a dark ring using the cobalt blue filter, it represents deposition of iron in the epithelium around the base of the cone of the cornea due to stagnated tears in that region. Vogts straie are verticle stress lines within the posterior stroma and the Descemet’s membrane of the cornea which occur due to severe stretching of the cornea tissue and often disappear with mild pressure or indentation of the cornea. Rizzuti’s sign is a conical reflection of light on the nasal cornea when light is shone from the temporal side, this deformation of the shape of the light is due to the conical shape of the cornea (Figure 2). Munson’s sign is a V shaped deformation of the lower eye lid on downward gaze when a patient with keratoconus looks downwards, the conically shaped cornea pushes down the lower eye lid.

While the aforementioned tools, techniques and signs do not take away the need for corneal tomography in the diagnosis and management of keratoconus, in situations where these expensive devices are not available, eye care practitioners should step up to the challenge with these inexpensive tools, techniques and signs in diagnosing this serious condition, preventing mis diagnosis or under-diagnosis of a condition with severe visual complications and impact on quality of life.
These inexpensive tests, techniques and signs can be carried out on patients with known risks for developing keratoconus, such as patients with vernal kerato-conjunctivitis, systemic allergic diseases associated with keratoconus, patients with a history of eye rubbing.
REFERENCES
1. Gokhale NS. Epidemiology of keratoconus. Indian J Ophthalmol. 2013;61(8):382.
2. Akowuah PK, Kobia-Acquah E, Donkor R, Adjei-Anang J, Ankamah-Lomotey S. Keratoconus in Africa: A systematic review and meta-analysis. Ophthalmic Physiol Opt. 2021;41(4):736–47.
3. Al-Mahrouqi H, Oraba SB, Al-Habsi S, Mundemkattil N, Babu J, Panchatcharam SM, et al. Retinoscopy as a Screening Tool for Keratoconus. Cornea. 2019;38(4):442–5.
4. Kennedy RH, Bourne WM, Dyer JA. A 48-year clinical and epidemiologic study of keratoconus. Am J Ophthalmol. 1986;101(3):267–73.
5. Zadnik K, Barr JT, Gordon MO, Edrington TB. Biomicroscopic signs and disease severity in keratoconus. Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study Group. Cornea. 1996;15(2):139–46.
6. Zadnik K, Barr JT, Gordon MO, Edrington TB, Group CS. Biomicroscopic signs and disease severity in keratoconus. Cornea. 1996;15:139–46.
7. Wagner H, Barr JT, Zadnik K. Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study: methods and findings to date. Cont Lens Anterior Eye. 2007;30(4):223–32.


